A Box of Snow
Posted by Faith BarbutoLast week we spent some outdoor time collecting a box of snow. We wondered what would happen if we took the snow inside, so we did. When we came in the next day, we found a box of water- dirty water! I reminded the students to think of this next time they are tempted to eat snow. We also noted that the tub of water was not full, even though it had been filled with snow. I explained that there is air trapped in the snow but not in between the water molecules. This week’s frigid cold gave us a new idea, what would happen to the water if we put it outside? We put it out and checked on it often, and it got cold but it wasn’t until the next morning that we discovered… a box of ice! The budding scientists were filled with more questions such as what would happen if we tried to write on it? We tried it! We knew that it would melt if we kept inside but how long would that take? By the end of the day it was almost all melted so of course, we decided to try to freeze it again. The next day we took out some thermometers to measure the temperature of the ice and water throughout the day. Just like the rest of us, it was freezing!!
Elephants Toothpaste
Posted by Joshua BartosiewiczAs 8th graders continue to dive deeper into Chemistry, students participated in a lab referred to as “Elephant’s Toothpaste.” Within this lab, each student mixed various substances and compounds to produce a chemical reaction which took only seconds to execute. The students were given the following materials; safety equipment, an erlenmeyer flask, empty beakers for measuring, food coloring, dish soap, hydrogen peroxide (12%), and potassium iodide mixed with water. To start the lab, students poured 100ml of hydrogen peroxide into the Erlenmeyer flask along with a drop of food coloring. Next, the young scholars added in a giant squirt of dish soap to the flask. The final step to initiate the reaction is to add 20ml of the potassium iodide and water mixture to the flask of peroxide, soap and food coloring. Not too long after everything is combined, the potassium iodide reacts with the hydrogen peroxide, causing the peroxide to lose an Oxygen atom; however, the soap helps to trap the Oxygen atom that is breaking free from the peroxide therefore causing foam to rapidly build up and bubble(which looks like toothpaste!). In this entertaining lab students observed an exothermic (gives off/releases heat) chemical reaction that was sure to spice up the learning environment.
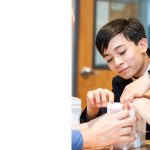
Slime Time!
Posted by Joshua Bartosiewicz- Making the perfect Slime!
- Mixing orange slime!
- Observing the reaction between mixing glue, saline, baking soda and shaving cream!
- Making the Slime fluffier by adding shaving cream.
- Checking out the change in consistency.
- “This was so cool!”
In 8th grade science, the students are continuing their exploration in the realm of Chemistry. Before leaving for winter break we were able to squeeze in a couple of exciting labs. Our young scholars are beginning to understand how substances are composed of molecules and atoms and that there are various states of matter (i.e. Solids, Liquids, Gases and Plasma). In this lab, students were able to see substances change phases; by mixing glue, contact saline solution, baking soda and shaving cream. The concoction resulted in a thick gelatinous substance referred to as “Slime”. Seeing these reactions in person really changes your perspectives on the subject matter. In the end, all of the students had a wonderful time and were even able to bring home their science experiments!
Plant Dissection!
Posted by Joshua Bartosiewicz- “This plant is made of a bunch of parts.”
- Checking out pollen under the microscope.
- Sorting the structures.
- Drawing and labeling the structures they’re investigating!
In the science lab, the 7th grade has been learning about plant and animal reproduction, heredity and growth. This lab was a plant dissection where the students were able to get a first hand look at the structures that make up an Easter Lily (flower). In this lab, students took apart the flower and sorted each structure. The young scholars were were asked to draw and label each structure, describe the patterns in the flower structures, and discuss the functions of the structures. Then they were asked what type of body features they’d expect an animal to have that pollinates that specific flower species. Throughout this lab, students had hand lenses and microscopes available to them to help them analyze each flower structure (being able to see pollen and other features on a microscopic level really changes your perspective on the natural world around us!). Overall, students had a blast investigating the parts and structures that help it to function and survive in an ever changing environment.
Can We Eat This?
Posted by Colleen NoyesCan we eat this? is the question MOST asked before many experiments. There will only be one time that this is true for the 6th grade! It is the plate tectonics experiment where students simulate the movement of the Earth’s plates using graham crackers (sometimes gluten free) and frosting (magma). Science can be a tasty learning experience…….but don’t ask me again.
In addition the 6th grade made posters representing the way that rocks and minerals are formed. Each group researched their topic and created a poster. They are posted on the bulletin board in the science hallway, check them out! Test your knowledge and take the quiz!
It’s been a busy fall –
Observing Carrot Organs!
Posted by Joshua Bartosiewicz- Slicing up the carrot to observe the organs and parts.
- Cutting up a carrot to see it’s organs.
- Checking out the carrot under a magnifying glass.
- An even closer look at carrot slices.
- Observing vascular and ground tissue of a carrot.
- Recording their observations.
The 7th graders have been studying the levels of organization within different organisms in science. We started out the school year learning about the basic building blocks of life known as cells. Once we learned about cell structures and their functions, we started looking at how these cells work together to help organisms perform life processes. The students began to see that cells are the foundation to organisms, but a collection of similar cells performing a similar task is known as “tissue”. Then, a collection of “tissue” is what makes up our “organs”, from there, a group of organs working together to perform a task are a part of an “organ system”. Finally, a combination of organ systems is what comprises an organism. In conclusion, the levels of organization is as follows; cells -> tissue -> organs -> organ systems -> organisms.
After students were able to grasp that concept, we started looking at the systems that make-up animal bodies (respiratory, circulatory, digestive, muscular systems, etc.) as well as plant bodies (shoot system, root system, etc.). In this lab, the students observed a carrot (which is a taproot to the carrot plant!) with and without a microscope. Our young scholars recorded their observations which included being able to see the vascular tissue and ground tissue of the carrot underneath the microscope. Upon completing this lab, the students were able to see real life examples of the finer details that comprise a plant body (a carrot in this case).