Elephants Toothpaste
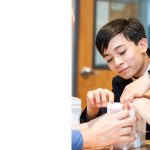
Posted by Joshua BartosiewiczAs 8th graders continue to dive deeper into Chemistry, students participated in a lab referred to as “Elephant’s Toothpaste.” Within this lab, each student mixed various substances and compounds to produce a chemical reaction which took only seconds to execute. The students were given the following materials; safety equipment, an erlenmeyer flask, empty beakers for measuring, food coloring, dish soap, hydrogen peroxide (12%), and potassium iodide mixed with water. To start the lab, students poured 100ml of hydrogen peroxide into the Erlenmeyer flask along with a drop of food coloring. Next, the young scholars added in a giant squirt of dish soap to the flask. The final step to initiate the reaction is to add 20ml of the potassium iodide and water mixture to the flask of peroxide, soap and food coloring. Not too long after everything is combined, the potassium iodide reacts with the hydrogen peroxide, causing the peroxide to lose an Oxygen atom; however, the soap helps to trap the Oxygen atom that is breaking free from the peroxide therefore causing foam to rapidly build up and bubble(which looks like toothpaste!). In this entertaining lab students observed an exothermic (gives off/releases heat) chemical reaction that was sure to spice up the learning environment.
← What Note is That?! Asexual and Sexual Reproduction Lab →